By guest author Dr Marcus J Head
It seems almost impossible to read news reports and social media posts these days without coming across someone telling you how important it is to look after your microbiome. From managing our mood and our weight, to reducing the risks of any number of diseases, someone, somewhere, will make a link to the billions of bacteria living in you, or on you.
But a recent study, published in the open-access journal Nature Scientific Reports, made a new and extraordinary connection: a foal’s internal hitchhikers may affect how fast he runs. And not just that – they can predispose to, or protect from, a range of conditions that might even prevent him from racing at all.
The paper followed 52 foals, born between February and May 2018, from five Thoroughbred stud farms in the UK. These foals were followed as they entered training (dispersed between 29 different training yards) and through to the end of their 3-year-old season.
In the earliest part of the study, faecal samples were obtained from each individual on day 2 postpartum and at regular intervals up until one year of age. These samples were then analysed and the number, variability, and different types of bacteria in the samples were assessed and recorded. The study was prompted by increasing evidence that not only are there a range of non-communicable diseases in humans affected by the bacteria within the intestines, but also that babies with differing birth and early-life circumstances may be predisposed to certain conditions, because of differences in their microbiome.
The clinical and racing histories of the foals were then compared to the results of the microbiological evaluations.
The results were striking: the gut bacterial community in the first months of life could predict the risks of specific diseases and athletic performance in later life. Foals with lower diversity (i.e. fewer different species of bacteria in their samples) at one-month-old had a significantly increased risk of respiratory disease in later life. A higher diversity was associated with a decreased risk of orthopaedic disease. And, when athletic performance was assessed — judged by comparing official ratings, number of starts/wins/places, and prize money earned — positive effects were again seen in foals with a greater diversity of bacteria at one-month-old and could even be linked to the relative abundance of specific bacterial families.
Foals are born without a functioning microbiome and must acquire all of their gut bacteria from their dam and their environment. This early phase of development, where foals are born with an immune system that is yet to encounter external challenges, is sometimes referred to as a ‘critical window’ — essentially a period when the gut microbial community imprints immunity and future health outcomes onto a blank canvas. There is now a great deal of interest in humans as to how this might be contributing to a variety of diseases: from obesity, diabetes and allergies, to diseases of the circulatory system, bones and neurodevelopment disorders such as autism.
This particular study did not examine the gut microbial profile of the mares delivering and rearing the foals. But the authors point out that the gut bacterial profile of human mothers is known to be a primary driver of gut colonisation in human infants. Indeed, in human medicine, there is increasing engagement of methods to optimise this ‘handover period’ in neonates. There is limited evidence for how feeding mares may influence foals in this early phase. But a recent study, that fed two groups of mares on the same stud farm separate and distinct rations, demonstrated a stark difference in failure of passive transfer — assessed by levels of IgG in the foals’ blood within twenty-four hours of birth — between the two sets. Other researchers have confirmed that the microbial population of foals varies between stud farms, most likely due to differences in management styles. Numerous studies in other species have demonstrated links between maternal nutrition and various measures of colostrum quality, and there is evidence that a pro-inflammatory profile is seen in colostrum produced by obese mares, along with reduced medium-chain saturated fatty acid levels (important as an energy source and in the development of gut microbiota and immunity).
So, what if the balance could be tipped back in favour of the newborn foal, simply by adjusting how the mare is fed? And what if this could also affect the vigour of our young athletes? The developmental origin of health and disease (DOHaD) — first advocated by David Barker in the early 1990s — seeks to understand how exposure to environmental factors during periods of high plasticity in the early stages of life (including the gestational period) influences the program for development and disease risk in the progeny, that extends well into adult life.
Before the middle of the twentieth century, the foetus was believed to be immune from the effects of the maternal environment. It is now understood that in laboratory animals and humans, which is where of course almost all of this research has been done, numerous external factors influence the health and development of the foetus before birth. Historically, the focus has been on those causing adverse effects, such as polycyclic aromatic hydrocarbons — found in vehicle exhaust fumes and cigarette smoke — alcohol, and certain notorious pharmaceuticals. Epidemiogical studies began to link certain aspects of development, some not immediately obvious at birth, with environmental factors that had not been appreciated earlier and may be influenced by microbiome effects. For example, having “furry pets” at home impacts meconium bacterial diversity, and children reared with domestic animals from infancy have reduced prevalence of allergic-related diseases; maternal consumption of artificial sweeteners produces profound metabolic changes and microbiome changes in mouse pups (with profiles similar to those seen in human obesity). ‘Barker’s theory’ on the foetal origins of adult disease considered three characteristics: latency, where the effects may not be apparent until much later in life; persistency, in which conditions affecting the foetus continue; and genetic programming, where specific genes are switched on due to the prenatal environment. Brief windows at the very start of development, when a small number of cells are dividing rapidly and vulnerable to a range of influences, drive the development program of the entire individual, all the way through to adulthood.
Much of the work on the transferability of a healthy microbial population from one individual to another was prompted by early observations, reported in 1958, that patients affected by a particularly unpleasant and intractable cause of severe diarrhoea showed drastic improvements following a novel treatment. Clostridioides difficile is an anaerobic bacteria (it thrives in low-oxygen environments), commonly found in the intestinal tract of normal people. It doesn’t cause any issues until it proliferates uncontrollably, and the commonest reason for this is when the other bacteria in the intestine ecosystem are knocked out, usually by the patient receiving antibiotics for another problem. ‘C. diff’ is the commonest cause of antibiotic-associated diarrhoea, and it is notoriously difficult to treat in some cases — it only responds to a select group of antibiotics, many cases remain refractory, and it has a fatality rate of up to 40%. Back in the 1950s, the doctors were utilising a therapy that had been written about in the fourth century: transplanting gut bacteria from a healthy individual into the affected patient. And they did so by taking faeces from those healthy volunteers and delivering it via an enema (the very earliest records describe it being given as a ‘soup’).
Faecal microbiota transplantation (FMT) is now used in cases of C. diff that do not respond to first-line treatments and is well established for this, with impressive success rates of 80-90%. It works because it restores the normal balance of gut microbes, which prevents the Clostridia from maintaining a hold on the intestinal environment and, basically, out-competes them for the resources available. But as well as this, restoring the regular bacteria found in a healthy gut revitalises the relationship between the host — us, or the animal — and the trillions of single cell organisms we live with.
Estimates of the numbers of bacteria within the body vary, but it’s safe to say it’s in the tens of trillions. It’s also safe to say that in most people the bacteria outnumber the human cells, in some cases by an order of magnitude. Whether the same is true of horses isn’t known, but considering the horse has evolved to rely almost entirely on bacterial activity within the intestine for its existence, it’s almost certain that the numbers involved are even more impressive. But the bacteria (and other microorganisms such as viruses and fungi) don’t just provide the necessary enzymes to digest food — they have evolved with the host and are absolutely vital for the development and maintenance of other functions, particularly the immune and nervous systems. Even within the intestine, it isn’t just a matter of dissolving the food as it passes through for the animal to absorb: the microbes are fundamental to those pathways developing adequately and operating effectively, from the earliest stages of life to the very last day of it.
The most striking examples of the absolutely cardinal role of bacteria in health and development can be seen in laboratory animals. Mice can be born and reared in sterile conditions, so that they develop without any (or very few) microbes in their environment and without the normal gut colonies they would usually possess. These so-called germ-free, gnotobiotic, or axenic mice do not develop normally. They can be fed the optimum diet and housed comfortably but they do not thrive: their immune and digestive systems are stunted, they do not gain weight normally, and they are vulnerable to a wide range of diseases. Even if introduced to ‘healthy’ microbial populations found in conventionally-reared animals they do not recover, most likely because they have missed the aforementioned, once-in-a-lifetime, critical window. Interestingly, germ-free mice can live longer than their colonised compatriots (if reared very carefully), which has recently raised important questions over the role of the microbiome in ageing and its implications for humans.
Mammals need microbes. So where does a new-born mouse, human, or foal get these essential organisms from and, knowing what we know about the dangers that some types of bacteria present, how do they get the right ones?
It is now established that babies born via caesarian rather than vaginal delivery are more at risk from a number of conditions, including inflammatory and neurodevelopment disorders, and diseases of the bowel, lungs and joints, so much so that the technique of ‘vaginal seeding’ has become a much-discussed technique. Much of the impact of surgical delivery has been attributed to alterations in the microbial community that the infant is exposed to in the very earliest stages of neonatal life. While caesarian delivery is a very much rarer event in horses, the effect of early exposure to the ‘correct’ microbiome (sometimes referred to as ‘pioneer colonisers’) has been demonstrated in other species. The hygiene hypothesis is a term used to describe the way that early exposure to particular microorganisms is protective, and that a lack of such exposure leads to poor immune tolerance. Unfortunately, this term has sometimes been misunderstood: it doesn’t mean hygiene is a bad thing — we all need protection from pathogens — and it certainly doesn’t imply that having more infections during early life is advantageous. But a broad exposure to a variety of certain microbes is an essential part of development.
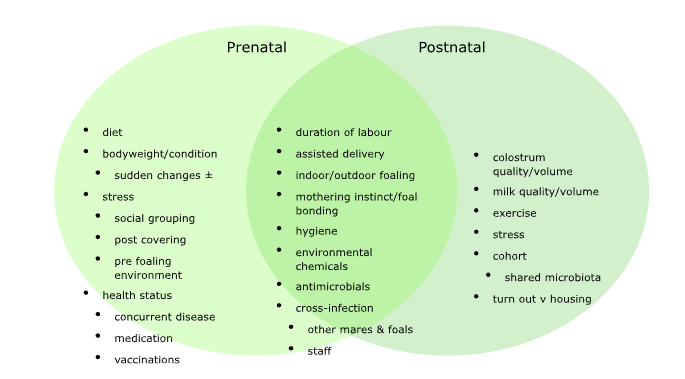
But this exposure may begin even earlier. For many years, it was considered incontrovertible that the foetal environment in utero was sterile and that exposure to bacteria and other organisms only began at birth. However, recent work has challenged this ‘sterile womb paradigm’ and a number of studies have demonstrated the presence of microbial material within placental and foetal tissues. The debate is still ongoing, not least around whether the presence of bacterial DNA actually represents viable bacteria or simply fragments of long-deceased organisms; even the ability to breed axenic animals (as mentioned above) suggests foetal microbial colonisation is unlikely.
What is emerging is a general consensus that the products of bacterial life in the mother may well be influencing foetal development, and could be crucial to it. Maternal microbiota produce bioactive metabolites that can enter the maternal circulation and may be actively or passively transferred through the placenta.
We know that elements of the microbiome that affect offspring are transferable: for example, bacteria taken from the gut contents of obese mice and transferred to pregnant, normal-weight mice, lead to overweight offspring with altered inflammatory responses. Maternal obesity causes changes in microbiota correlated with altered foetal brain development, while maternal undernutrition in humans affects infant immunity by attenuating responses to vaccines in early childhood (among many other effects). There is a huge volume of data concerning nutrition, and its effects on offspring, across many species, and it’s beyond the scope of this article to go into direct nutrient effects. Nonetheless, work has shown that foals born to obese mares are more likely to be affected by osteochondrosis, and supports the hypothesis that maternal relative hyperinsulinaemia may be a contributory factor through its role in cartilage differentiation. Foals born to overweight dams also have evidence of systemic inflammation (higher levels of plasma SAA) which may also affect development through inhibitory effects on bone growth. Crucially, the impact on progeny may be different depending on the alignment of the disturbance: i.e. whether the mares were obese at conception or overfed during the early or later stages of gestation. Maternal undernutrition is much less likely to be a problem in commercial breeding operations, but when it is, mares may develop a syndrome close to gestational diabetes in humans, a condition widely recognised to have potentially very serious consequences to the neonate, and even beyond. Again, timing is crucial: studies of epidemics, famine, and war show that mothers and their children have different outcomes depending on at which stage, from preconception to early childhood, the crisis occurred.
Maternal stress during early pregnancy has been shown to reduce faecal bacterial diversity and alter taxa composition in pregnant mice and has been replicated in human studies. Conversely, severe maternal stress in the last trimester was associated with increased bacterial diversity in newborn babies. Maternal depression may alter the ratio of certain bacterial families in neonates, specifically by reducing numbers of Proteobacteria, a pioneer taxon which creates the environment for later anaerobic colonisation.
A number of studies have identified known risk factors for infant severe respiratory tract infections, the leading cause of child mortality globally, including a carbohydrate-rich, fruit-/fibre-/vegetable-poor maternal diet, and/or never breastfeeding. In a 2023 study, researchers demonstrated that mice fed high-fibre diets produced a milk microbiota that increased infant gut propionate levels, leading in turn to increased production of specific cells in the bone marrow that confer protection against respiratory infections. Indeed, therapy with specific propionate-producing bacteria, and even propionate supplementation, were shown to act in a similar way. Fermentable dietary fibre favourably influences the gut microbiome resulting in the production of metabolites that modulate the immune system (particularly short-chain fatty acids — SFAs). It is widely accepted that SFAs play an important part in regulating intestinal barrier integrity, and there is now evidence that they have a role in placental structure and vascularisation. Breastmilk also contains regulatory cytokines, growth factors and oligosaccharides, all of which influence the developing infant. Metabolic disorders, such as gestational diabetes, affect the levels of SCFAs and other metabolically significant metabolites causing changes in the maternal immune system with knock-on effects for the developing foetus. The authors of this study suggested that the “manipulation of the maternal diet during pregnancy and during the pre-weaning period, or through the provision of defined tailor-made prebiotics and/or probiotics directly to infants, may present novel opportunities for enhancing resistance to inflammation-mediated diseases.” Other researchers have described the transport of even more molecules across the placenta, including lipopolysaccharides and other bacterial cell fragments. Preconception infections with some intestinal parasites can lead to maternal imprinting in mouse offspring, thereby suggesting that the transfer of immunity is not solely dependent on the passive transfer of antibodies.
These metabolites may also exert an effect on the genetic material within cells. Epigenetics is the set of factors and molecules that modify gene expression while not changing the genome sequence itself. Only around 2% of mammalian DNA is associated with protein-encoding genes; the rest (non-coding DNA — what used to be referred to as ‘junk DNA’) modifies the links on the chain to switch genes on and off at crucial junctures. These modifications are commonly associated with environmental cues — good and bad — that can result in significantly different outcomes (phenotypic plasticity) for two genetically identical individuals. In one Danish study, children who exhibited allergic rhinitis at the age of six were found to have a distinct DNA methylation pattern (an epigenetic signature) in their nasal mucosal cells associated with reduced microbial diversity at one week of life. It was folate’s role in countering the development of neural tube defects that first pointed to the foetal nutritional programming hypothesis through epigenetic programming. And as the stage most susceptible to environmental stressors is embryogenesis, where DNA methylation is closely associated with optimal differentiation and growth, consideration of preconception dietary factors is fundamental (folate supplements are recommended when pregnancy planning, but gut bacteria produce significant amounts of folate in humans with a healthy microbiome).
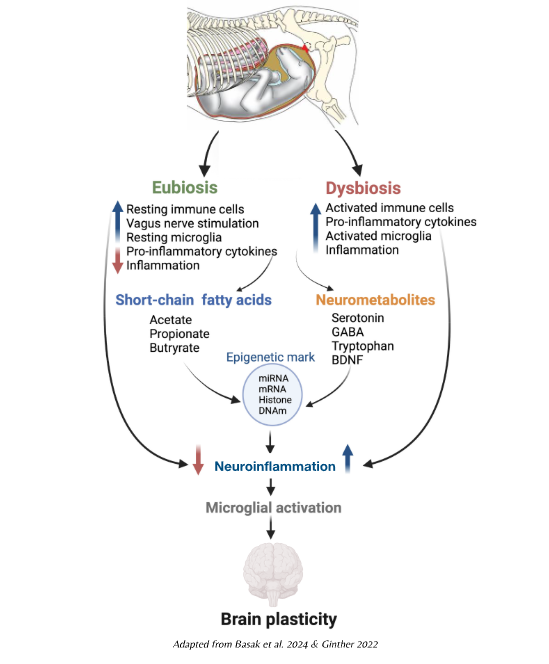
While epigenetic changes do not change the base-pair sequences within the genome, they are heritable, meaning that future generations may inherit far more than just the A–T and G–C building blocks. It seems certain that different epigenetic mechanisms may partly explain why dietary factors at critical developmental stages might affect the susceptibility to diseases in adulthood.
With microbes contributing 99% of all genetic information present in the body, maternal microbial transmission should be viewed as an additional and important mechanism of genetic and functional change in evolution. Horses are selected for characteristics such as temperament, strength, speed, and endurance. Although genetic selection tools have been developed to assist breeders in the choice of sires and dams, there is also a growing body of research reporting that the maternal environment modulates the genetic potential of the offspring. Under natural conditions, microbes that promote host fitness, especially in females, will simultaneously increase their odds of being transferred to the next generation. Under controlled breeding programmes, efforts to assist this beneficial effect should be encouraged: the trajectory of the young foal’s colonisation process has lifelong clinical implications. And being aware of its influence may not only improve the outlook for your foal, but your foal’s foals as well.





